Calcium #2 is a photograph by Science Photo Library which was uploaded on October 3rd, 2018.
Calcium #2
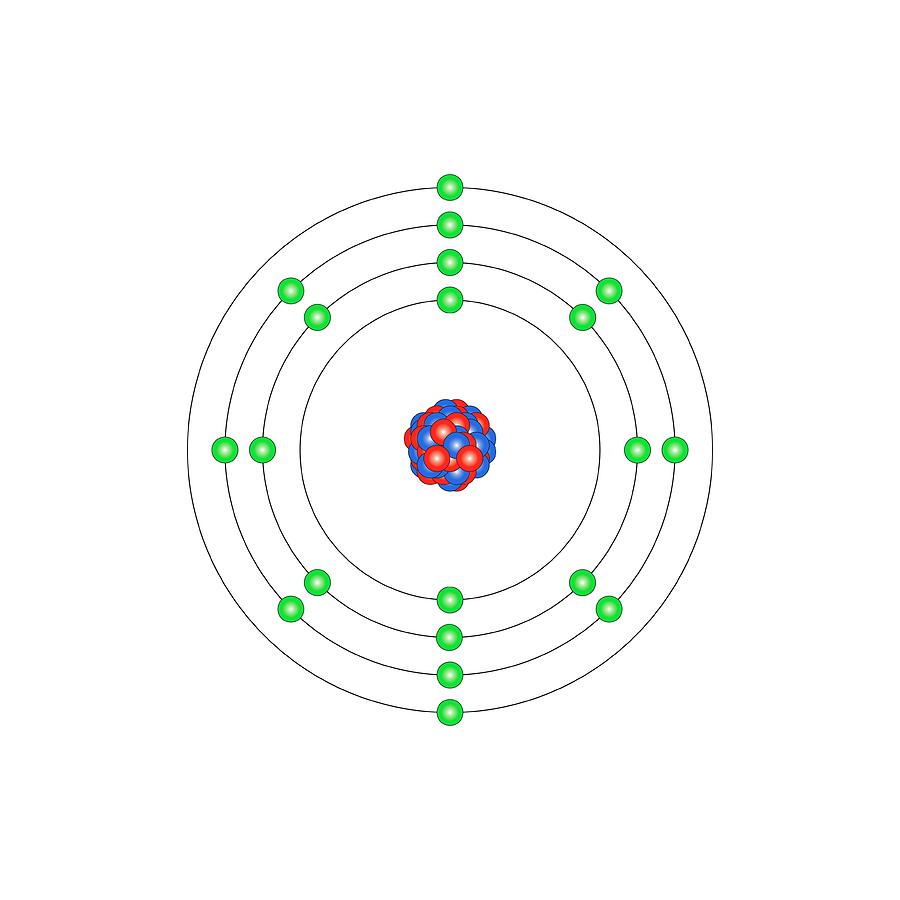
Calcium (Ca). Diagram of the nuclear composition and electron configuration of an atom of calcium-40 (atomic number: 20), the most common isotope of... more
Title
Calcium #2
Artist
Science Photo Library
Medium
Photograph
Description
Calcium (Ca). Diagram of the nuclear composition and electron configuration of an atom of calcium-40 (atomic number: 20), the most common isotope of this element. The nucleus consists of 20 protons (red) and 20 neutrons (blue). 20 electrons (green) bind to the nucleus, successively occupying available electron shells (rings). The stability of an element's outer electrons determines its chemical and physical properties. Calcium, vital for cellular function, is an alkaline earth metal in group 2, period 4, and the s-block of the periodic table. In elemental form, it is a reactive metal that burns with a bright orange-red flame when heated in air.
Uploaded
October 3rd, 2018
More from Science Photo Library
Comments
There are no comments for Calcium #2. Click here to post the first comment.


































